what makes a cation less likely to form
Understanding The 7 Key Factors That Stabilize Negative Charge (And Ultimately, Basicity)
Similar I wrote virtually last time, it's expert – simply not enough – to recognize partial charges and to effigy out where they interact.
Since reactions involve processes that lead to the gain or loss of charges, understanding the factors that stabilize (or destabilize) accuse have a tremendous impact on how likely a reaction is given to occur! Allow's talk virtually negative charge today, which mostly means "anions" for our purposes but tin can broadly refer to any lone pair of electrons.
Table of Contents
- Agreement The Stability Of Negative Charge Helps Us Understand What Reactions Are Likely (And Not Likely) To Occur
- Cistron #one: High Charge Densities Are Unstable
- Factor #2: As Electronegativity Increases Beyond The Periodic Table, So Does The Stability Of Negative Accuse
- Cistron #3: As Polarizability Increases Down The Periodic Tabular array, So Does The Stability Of Negative Charge
- Cistron #4: Resonance Stabilization Of Negative Charge
- Gene #5: Electron-Withdrawing Groups (Anterior Effects) Stabilize Negative Charge
- Increasing s-Character In The Hybridization Of An Atom Is Effectively Similar Increasing Its Electronegativity
- A Special Instance: Aromaticity
- "Basicity" Is Only Another Word For "Stability Of The Negative Charge"
1. Understanding The Stability Of Negative Charge Helps Us Sympathise What Reactions Are Likely (And Non Likely) To Occur
Permit's talk about a concrete example. For instance if a reaction leads to the formation of a very unstable negative charge, it's unlikely to occur. Simply if information technology leads to the loss of a very unstable negative charge, information technology's considerably more likely.
For instance, that's why 1 of these reactions of marsh gas is likely and the other is unlikely. These acid-base of operations reactions are covered here, BTW.
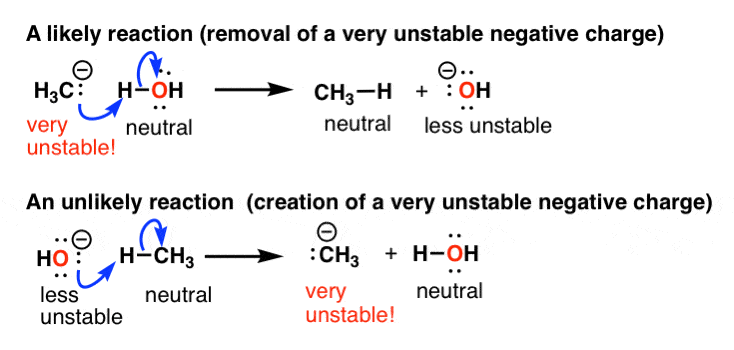
So what factors lead to the stabilization of negative accuse? Two main things.
- negative charge is stabilized by adjacent positive charge (opposite charges attract!)
- negative charge tends to exist less stable when it'due south concentrated and more stable when information technology's dispersed.
Remember about that as you look at this list of seven factors that stabilize negative charge.
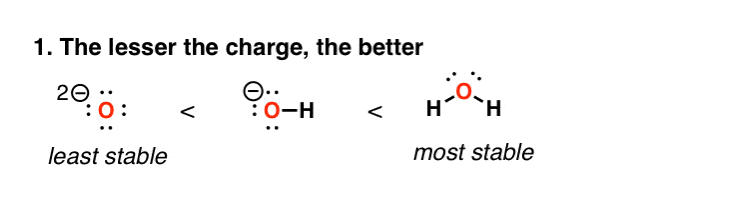
two. Factor #ane: Loftier Charge Densities Are Unstable
This one's fairly straightforward to understand. High charge densities are unstable. So as we motility from water to HO(–) to O(2-), we are getting progressively more than unstable here. This is reflected in acerbity tables; water (H2O) is far more probable to lose a proton to give HO(–) than HO(–) is likely to lose a proton to give O(2-)
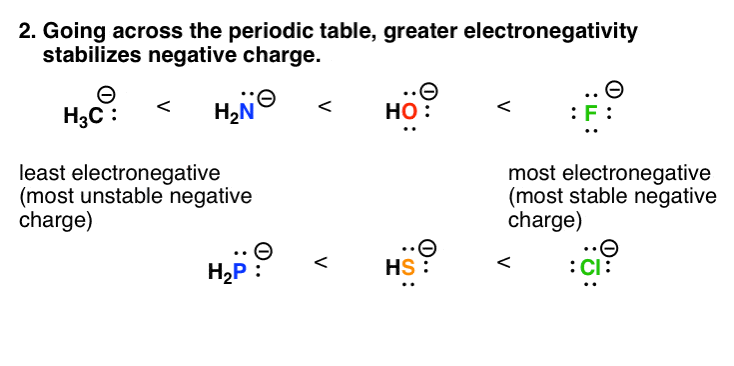
3. Factor #two: As Electronegativity Increases Across The Periodic Table, And then Does The Stability Of Negative Charge
Electronegativity is a crude measure how effectively the positively charged nucleus of an atom can "pull" electrons toward information technology. (Opposite charges concenter.) Electronegativity increases equally nosotros go across the periodic table. Then if you lot compare the anions going from C , N, O to F beyond the periodic table, the stability of the negative charge will increase. This is likewise reflected in acidity values.
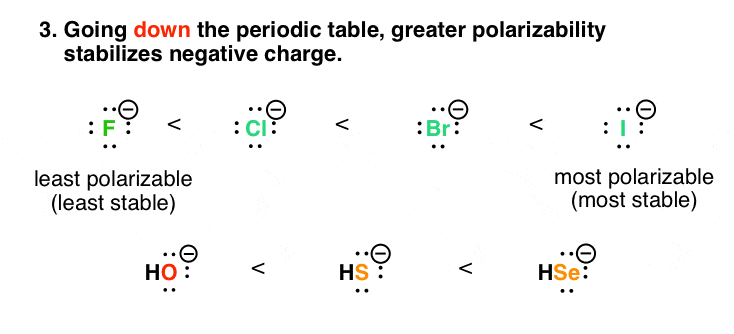
4. Factor #3: Equally Polarizability Increases Down The Periodic Table, So Does The Stability Of Negative Charge
Downward the periodic table, it's a lilliputian more helpful to think "dispersal of charge is good!" rather than "contrary charges attract". Compare fluorine and iodine. The size of the fluorine ion (radius: 119 pm) is much smaller than iodine (radius: 206 pm). Still, they both accept a charge of negative one.
Imagine ii balls, each weighing one pound. But ane is made of fe, and the other is made of rubber. Which ball is going to be smaller? The iron ball (smaller and harder – more dumbo) is like fluorine, and the condom ball (larger and squishier) is like iodine. And a certain "squishiness" helps to stabilize charge, since it isn't as full-bodied over a minor volume. That's a way of expressing the greater polarizability of iodine. Depression accuse densities are more stable!
five. Factor #four: Resonance Stabilization Of Negative Charge
Forth the aforementioned lines, a negative charge that is next to one or more Pi bonds tin disperse its negative accuse over multiple atoms. We depict this phenomenon as "resonance". So in the case below, the negatively charged methane series on the left is much less stable than the next negatively charged species, where the negative charge can be dispersed over multiple carbons through resonance. This is another case of how reducing charge density (or spreading it out) is a stabilizing influence.
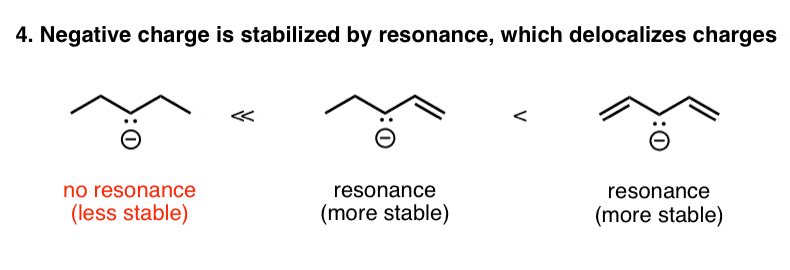
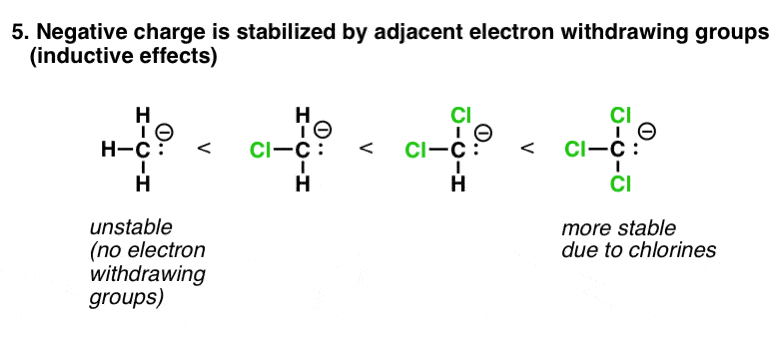
6. Factor #5: Electron-Withdrawing Groups (Inductive Effects) Stabilize Negative Charge
This i falls more into the auspices of "contrary charges attract". A negative accuse that is adjacent to an cantlet with electron withdrawing groups on it will be much more stable than an equivalent atom that is non. In the extreme case of CCl3(-), the resulting ion is many orders of magnitude more than stable than HiiiC(-) itself. (This is the basis of the haloform reaction).
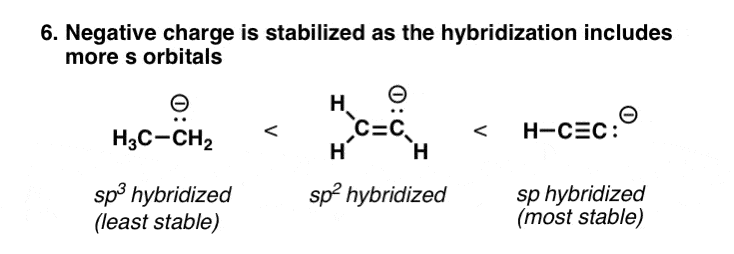
seven. Increasing southward-Graphic symbol In The Hybridization Of An Cantlet Is Effectively Like Increasing Its Electronegativity
s orbitals are closer to the nucleus than p orbitals are. So electrons that are in s orbitals will be closer to the nucleus than electrons in p orbitals – and therefore, lower in free energy ("contrary charges attract"). For this reason, electrons that are in sp orbitals are lower energy than spii, which is lower energy than sp3, since they accept greater s character (33% for sptwo) than sp3 (25%). This makes the anions more than stable, simply as if it were on a more electronegative atom.
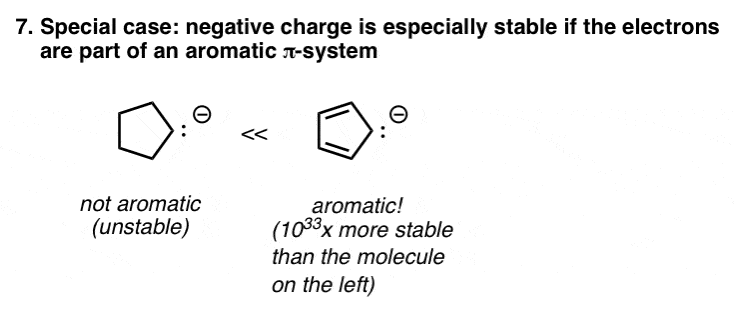
8. A Special Case: Aromaticity
This is a special case, covered in detail in organic chemistry 2 (peek alee). Certain molecules possess a special stability – called aromaticity – that is enormously stabilizing, kind of like qualifying for a huge revenue enhancement interruption from the government. Certain negatively charged molecules – such as the cyclopentadienyl anion, pictured beneath – are aromatic, and therefore possess much greater stability than they would accept otherwise.
nine. "Basicity" Is Just Another Word For "Stability Of The Negative Accuse"
Seven factors?!!! So how do we know which is almost important? That'south a great question! These trends tin interact with each other in unpredictable ways, and it's hard to judge which is most important.
Thankfully, there's a concept you lot've probably already met for figuring out the stability of these species, which can be readily measured. It'south chosen basicity. These factors determine how stable a base will be!
The basicity of a species tells you nigh how stable its lone pair of electrons are.
How do we find a good measure out of basicity? Simple. It's in the pKa table, a drove of measurements that's been compared to the tabular array of manus strengths in poker.
Bottom line:
- Two factors to lookout man out for: opposite charges attract, and dispersal of charge.
- unstable anions will tend to be at the intial tails of arrows (form bonds).
- stable anions will tend to be at the terminal heads of arrows (probable to be leaving groups)
Source: https://www.masterorganicchemistry.com/2012/02/27/7-factors-that-stabilize-negative-charge-in-organic-chemistry/
Post a Comment for "what makes a cation less likely to form"